No products in the cart.
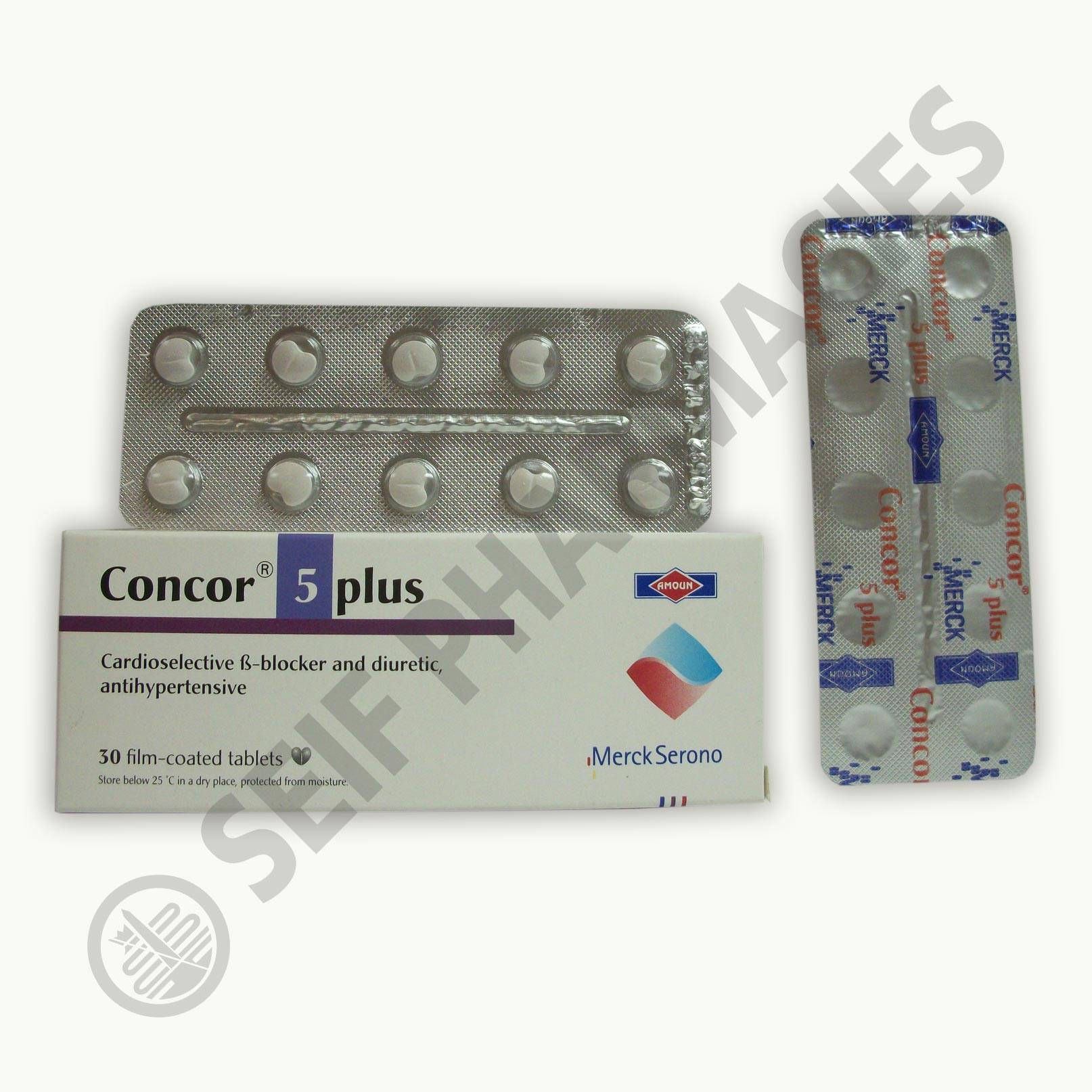
** Concor® 5 plus 30 film-coated tablets. Cardioselective beta-blocker and diuretic, antihypertensive.
** Composition:
1 film-coated tablet contains:
Bisoprolol fumarate (2:1) 5 mg and Hydrochlorothiazide 12.5 mg
** Indications:
– Arterial hypertension.
– The combination preparation Concor 5/10 plus is recommended if monotherapy is not sufficiently effective.
** Contra-indications:
– manifest heart failure.
– shock.
– 2nd or 3rd degree AV block.
– sick sinus syndrome.
– sinoatrial block.
– bradycardia (resting pulse less than 50 beats/min before the start of therapy).
– acidosis.
– bronchial hyperresponsiveness (e.g. bronchial asthma).
– late stages of peripheral circulatory disturbances.
– concomitant administration of MAO inhibitors (exception: MAO-B inhibitors).
– severe disturbances of kidney function (renal insufficiency with oliguria or anuria; creatinine clearance less than 30 mL/min and/or serum creatinine more than 1.8 mg/100mL.
– acute glomerulonephritis.
– coma and hepatic precoma.
– refractory hypokalaemia.
– severe hyponatraemia.
– hypercalcaemia.
– gout.
– hypersensitivity to hydrochlorothiazide and other thiazides, sulphonamides, and beta-blockers.
– children (no therapeutic experience).
** Special warnings and precuations for use:
Patients with any of the following should be monitored closely:
– 1st degree AV block.
– mainfest or latent diabetes mellitus (severe hypoglycaemic conditions possible; regular monitoring of blood glucose).
– prolonged periods of strict fasting and heavy physical strain (possibility of severe hypoglycemic conditions).
– patients with phaeochromocytoma (tumor of the adrenal medulla) (Concor plus should not be administered until after alpha-blockade).
– hypovolaemia.
– cerebral sclerosis.
– coronary sclerosis.
– impaired kidney function of mild degree and co-existing impairment of liver function (dose adjustment).
– Prinzmetal’s angina.
– In patients with psoriasis in their psersonal or family history medication containing beta-blockers should only be prescribed if the risk/benefit has been carefully evaluated.
– Beta blockers can increase the sensitivity to allergens and the severity of anaphylactic reactions. Therefore, Concor plus should only be prescribed if considered essential in patients with a history of severe hypresensitivity reactions and in patients undergoing desensitisation therapy (beware of of excessive anaphylactic reactions).
** Psology and method of administration:
– The treatment of high blood pressure should principally be started with low doses of a single active substance and then increased gradually.
– The fixed combination of Concor 5/10 plus consisting of bisoprolol and hydrochlorothiazide should be administered only if the blood pressure could not be normalised by the individual active substances or else excessive adverse reactions had occured at high doses. The following apply as guidelines:
# Arterial hypertension:
– Normally 1× 1 film-coated tablet of Concor 5 plus (equivalent to 5 mg bisoprolol and 12.5 mg hydrochlorothiazide) daily. If the blood pressure is only inadequately reduced the dose may be increased to 1 × 2 film-coated tablets of Concor 5 (equivalent to 10 mg bisoprolol and 25 mg hydrochlorothiazide) or 1 film-coated tablet of Concor 10 plus once daily. In co-existing impairment of kidney or liver function, the elimination of the HCT component of Concor 5/10 plus is reduced, so that preference may be to the lower dose form.
# Mode and duration of administration:
– The film-coated tablets are to be swallowed whole with the breakfast with some liquid.
– There is no limit to the duration of administration. It depends upon the nature and severity of the disease.
– After long-term therapy – particularly in the presence of ischaemic heart disease Concor 5/10 plus should be discontinued gradually (i.e. over 7-10 days), since an abrupt withdrawal may lead to an acute deterioration of the patient’s condition.
** Dosage as directed by the physician. By prescription only.
** Store below 25 degrees C, in a dry place, protected from moisture.
** Manufactured by: AMOUN PHARMACEUTICAL CO. S.A.E. – El-Obour City, Cairo, Egypt
Under licence from: Merck KGaA – Darmstadt – Germany (Merck Serono)
To be sold in Egypt only
| Weight | 100 kg |
|---|
Add a review
You must be logged in to post a review
Log In
Q & A
There are no questions yet
Ask a question
Your question will be answered by a store representative or other customers.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Thank you for the question!
Your question has been received and will be answered soon. Please do not submit the same question again.
Error
An error occurred when saving your question. Please report it to the website administrator. Additional information:
Add an answer
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Thank you for the answer!
Your answer has been received and will be published soon. Please do not submit the same answer again.
Error
An error occurred when saving your answer. Please report it to the website administrator. Additional information:














9 reviews for CONCOR PLUS 5 MG 30 TAB