No products in the cart.
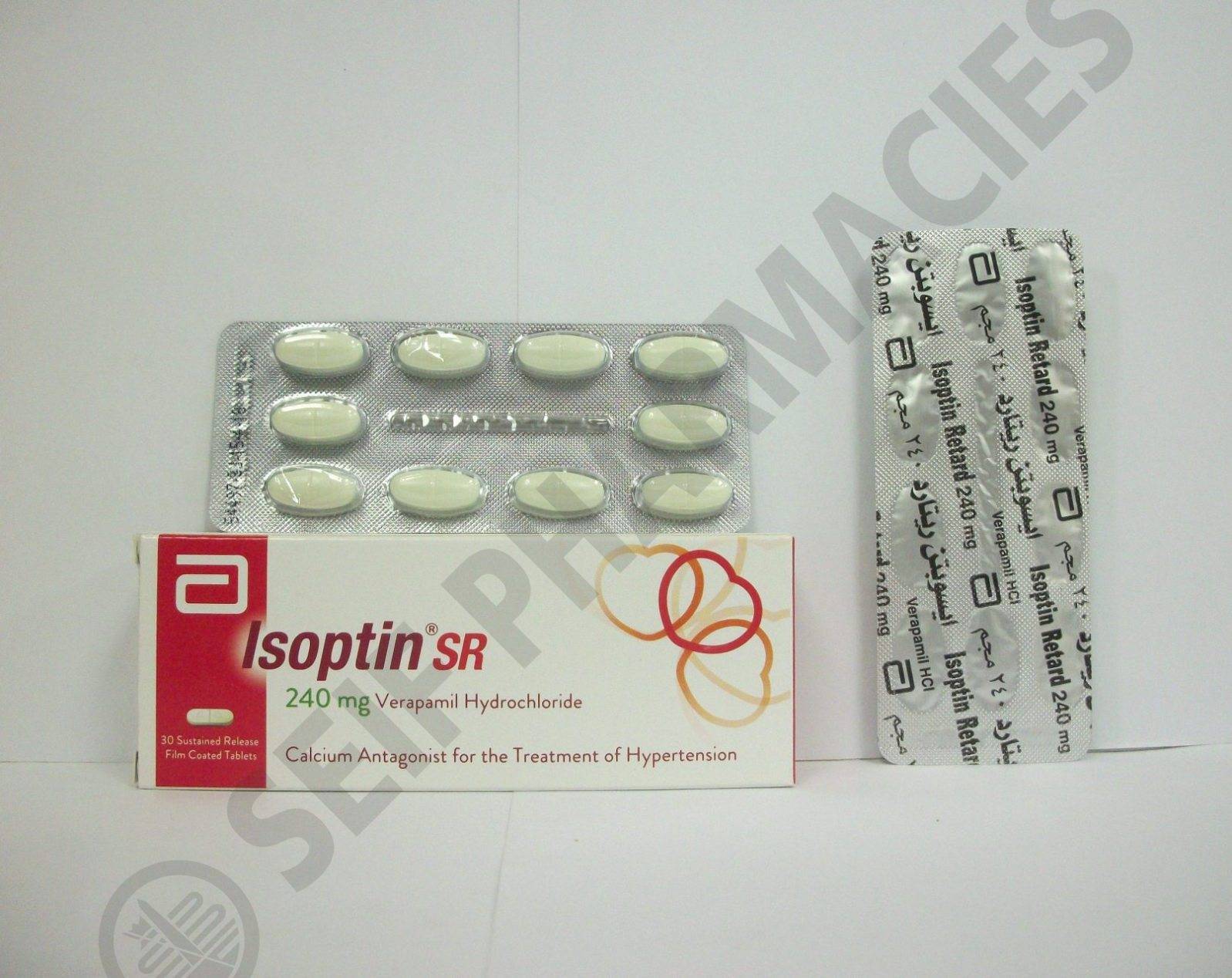
** Isoptin SR (240 mg Verapamil hydrochloride) 30 Sustained-Release Film-Coated Tablets.
Calcium antagonist (cardioselective) for the Treatment of Hypertension.
** Composition:
Each sustained-release film-coated tablet contains:
Verapamil Hydrochloride 240 mg
** Indications:
– Hypertension.
– Secondary prevention after acute myocardial infarction.
** Dosage and mode of administration:
– The dose of Isoptin SR 240 mg, individualized according to the severity of the disease are to be taken regularly as prescribed by the physician.
– The film coated tablets are to be swallowed whole with some liquid, preferably with or shortly after meals.
– Unless otherwise instructed, the daily dose is 1 film coated tablet in the morning (patients requiring particularly gradual blood pressure lowering should be started on half a tablet taken in the morning).
– If after about 2 weeks of treatment a dose increase is found to be necessary, the dose can be raised to a maximum of 2 film coated tablets daily.
– On long-term treatment, a daily dose of 480 mg should not be exceeded.
– Short-term dose increases are possible only when directed by the physician.
– In patients with impaired hepatic function, the effect of Verapamil is intensified and prolonged depending on the severity of the liver disease due to diminished drug metabolism. In these cases, dosage should be adjusted with special care starting with the lowest possible dose.
** Contraindications:
– Cardiovascular shock.
– Complicated acute myocardial infarction (bradycardia, marked hypotension, left ventricular failure).
– Severe conduction disorders (2nd and 3rd degree AV block).
– Sick sinus syndrome (bradycardia, tachycardia syndrome).
** Warnings in the leaflet must be revised.
** Store at a temperature not exceeding 30 degrees C, away from light.
Keep out of reach of children.
** Manufactured by: Kahira Pharmaceuticals && Chemical Industries Company
Under License of: Abbott Laboratories – USA && its subsidiary in Germany
| Weight | 100 kg |
|---|
Add a review
You must be logged in to post a review
Log In
Q & A
There are no questions yet
Ask a question
Your question will be answered by a store representative or other customers.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Thank you for the question!
Your question has been received and will be answered soon. Please do not submit the same question again.
Error
An error occurred when saving your question. Please report it to the website administrator. Additional information:
Add an answer
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Thank you for the answer!
Your answer has been received and will be published soon. Please do not submit the same answer again.
Error
An error occurred when saving your answer. Please report it to the website administrator. Additional information:

















2 reviews for ISOPTIN SR RETARD 240 MG 30 TAB