لا توجد منتجات في سلة المشتريات.
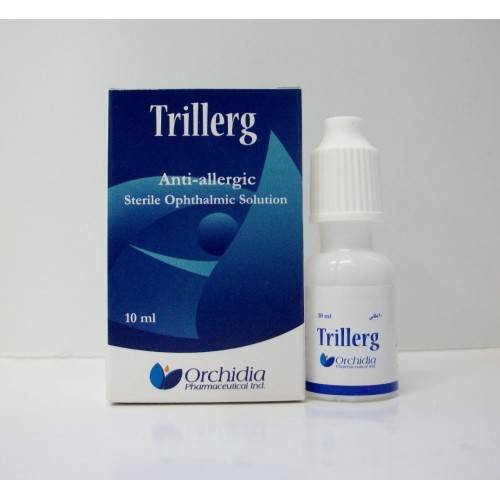
| * Trillerg Sterile Ophthalmic Solution 10 ml. Anti-allergic. | ||
| * Composition: Each mL contains: | ||
| – Active: Antazoline HCl 0.5 mg + Tetryzoline HCl 0.4 mg. | ||
| – Preservative: Benzalkonium chloride 0.05 mg. | ||
| – Excipients: Hydroxy propyl methyl cellulose 3 mg + Diluted hydrochloric acid (HCl) q.s + Water for injection complete to 1 mL. | ||
| * Indications: | ||
| & Non-infectious irritative conjunctivitis, allergic inflammatory affections of the conjunctiva, hay fever | ||
| & and conjunctivitis vernalis. | ||
| & Apply 1 drop every 3 hours during the acute phase, for maintenance therapy 1 drop 2-3 times | ||
| & per day is sufficient. 1-2 drops per day are sufficient for children. | ||
| * Contra-indications: | ||
| – Hypersensitivity to substances contained in Trillerg. | ||
| – Patients with glaucoma caused by a narrow iridocorneal angle (narrow angle glaucoma) as well as | ||
| & in case of (dry eye) syndrome. | ||
| – Small children under the age of 2 years. | ||
| * Precautions: | ||
| – Use carefully with children. | ||
| – Other therapeutic measures or medications should be chosen in case of chronic recurring allergies. | ||
| & The preparation is not intended for long-term medication. If a teatment longer than 2-3 days is | ||
| & forseen, it should be prescribed and monitored by a physician. | ||
| – Care is indicated in the case of rhinitis sicca. Due to systemic sympathomimetic effects care should | ||
| & be exercised when treating diabetics or patients suffering from severe heart disease, hypertension, | ||
| & hyperthyroidism or pheochromocytoma with the preparation. | ||
| – Eye infections can be masked. | ||
| – Contact lenses should be removed before the administration of the product. Thereafter wait at least | ||
| & 15 minutes before placing the lenses back into the eye. Trillerg may temporarily reduce tear production, | ||
| & resulting in reduced tolerance for contact lenses. | ||
| * Pregnancy / Lactation: | ||
| & Trillerg should be given to a pregnant or nursing woman only if clearly needed and caution should be | ||
| & exercised when administered. It is not known whether antazoline or tetryzoline is transferred into | ||
| & mother’s milk. (Pregnancy category C). | ||
| * Overdosage: | ||
| & Overdosing is not expected from the correct ophthalmic use of the preparation. Accidental ingestion does | ||
| & not lead to severe effects in adults. On the other hand, nausea, somnolence, arrhythmia/tachycardia | ||
| & and possibly shock may occur in children, in particular in infants under 2 years. | ||
* Prescription only medicine. For external use only. Description and Directions :
منتجات ذات صلة |