المجموع: 11.50 EGP
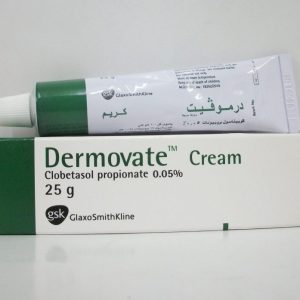
* Dermovate (Clobetasol Propionate 0.05%) ointment 25 g.
* Composition: Each 100 gm contains:
Clobetasol 17 propionate 0.054 gm equivalent to Clobetasol propionate 0.05 gm.
* Therapeutic indications:
– Psoriasis (excluding widespread plaque psoriasis).
– Recalcitrant eczemas.
– Lichen planus.
– Discoid lupus erythematosus and other skin conditions which do not respond satisfacorily to less active steroids.
* Dosage and administration:
Apply sparingly to the affected area once or twice daily. Therapy should be discontinued when control is achieved. Apply sparingly to the affected area once or twice daily. Therapy should be discontinued when control is achieved. courses of Dermovate may be used to control exacerbations. If continuous steroid treatment is necessary, a less potent preparation should be used. In very resistant lesions, especially where there is hyperkeratosis, the anti-inflammatory effect of Dermovate can be enhanced, if necessary, by occluding the treatment area with polythene film. Overnight occlusion only is usually adequate to bring about a satisfactory response. Thereafter improvement can be usually be maintained by application without occlusion.
* Contra-indications:
– Rosacea.
– Acne vulgaris.
– Perioral dermatitis.
– Perianal and genital pruritus.
– Primary cutaneous viral infections (e.g., herpes simplex, chickenpox).
– Hypersensitivity to the preparation.
– The use of Dermovate skin preparations is not indicated in the treatment of primary infected skin lesions caused by infection with fungi or bacteria; dermatoses in children ubder one year of age, including dermatitis and napkin eruptions.
* For external use only.
* Store at room temperature.
* Manufactured by: SmithKline Beecham (SB), Haram – Giza. For: GlaxoSmithKline (gsk) S.A.E. – Egypt.
Under License from: Glaxo Wellcome – UK.
DERMOVATE is a trade mark of the GlaxoSmithKline group of companies.
| الوزن | 100 كيلوجرام |
|---|

 BETADERM 0.1% 15 GM CREAM
BETADERM 0.1% 15 GM CREAM