Subtotal: 71.00 EGP
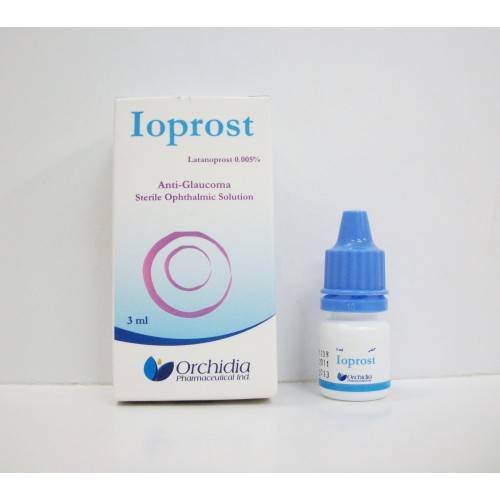
| * Ioprost (Latanoprost) 0.005% (50 mcg / ml) Sterile Ophthalmic Solution 3 ml. Anti-Glaucoma. |
| * Composition: Each ml contains: |
| & Latanoprost 50 mcg (0.005%). Benzalkonium chloride 0.2 mg. |
| * Indications and Usage: |
| & Ioprost Sterile Ophthalmic Solution is indicated for the reduction of elevated intraocular pressure |
| & in patients with open-angle glaucoma or ocular hypertension. |
| * Dosage and Administratiopn: |
| & The recommended dosage is 1 drop (1.5 mcg) in the affected eye(s) once daily in the evening. |
| & The dosage of Ioprost should not exceed once daily since it has shown that more frequent |
| & administration may decrease the intraocular pressure lowering effect. Reduction of the intraocular |
| & pressure starts approximately 3-4 hours after administration and the maximum effect is reached |
| & after 8-12 hours. Ioprost may be used concomitantly with other topical ophthalmic drug products |
| & to lower intraocular pressure. If more than one topical ophthalmic drug is being used, the drugs |
| & should be administered at least 5 minutes apart. |
| * Patients information: |
| – This product is sterile when packaged. Do not allow the dropper to touch eyelids or any other surface |
| & as this may contaminate the solution. |
| – Patients should be advised that if they develop any ocular reactions, particularly conjunctivitis and |
| & lid reactions or any ocular condition (e.g. trauma or infection), they should immediately seek physician’s advice. |
| – Contact lenses should be removed prior to administration of the solution. Lenses may be reinserted |
| & 15 minutes following administration of Ioprost. |
| * Pregnant and nursing mothers: Teratogenic Effects, Pregnancy Category C. |
| & Ioprost should be given to a pregnant or nursing woman only if clearly needed and caution should be exercised when administered. |
| * Pediatric and Geriatric Use: |
| & Safety and effectiveness in pediatric and geriatric patients have not been established. |
| * Prescription (Rx) only medicine. For external use only. |
| * Storage: Protect from light. Store unopened bottle(s) in refrigerator at temperature between (2 – 8 degrees C). |
| & During shipment to the patient, the bottle may be maintained at temperatures up to 40 oC for a period not exceeding 8 days. |
| & Once a bottle is opened for use, it may be stored at room temperature up to 25 degrees C for 6 weeks. |
| * Don’t use longer than 1 month from opening (Ioprost must not be used after 4 weeks from opening the bottle). |
| * Manufactured by: Orchidia Pharmaceutical Industries – Egypt. |
| Weight | 100 kg |
|---|
Q & A
Ask a question
Your question will be answered by a store representative or other customers.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Thank you for the question!
Your question has been received and will be answered soon. Please do not submit the same question again.
Error
An error occurred when saving your question. Please report it to the website administrator. Additional information:
Add an answer
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Thank you for the answer!
Your answer has been received and will be published soon. Please do not submit the same answer again.
Error
An error occurred when saving your answer. Please report it to the website administrator. Additional information:

 TWINZOL EYE DROPS 5 ML
TWINZOL EYE DROPS 5 ML 













Reviews
There are no reviews yet